|
 |
- Search
| Arch Aesthetic Plast Surg > Volume 25(3); 2019 > Article |
|
Abstract
The removal of fillers used for soft-tissue augmentation is an issue of concern, as the possible need for extensive surgery to remove fillers deters their use by many surgeons. Several studies have demonstrated the safety and efficacy of polyacrylamide hydrogel (Aquamid) gel, but to date no report has described its removal after 10 years. Here, we report a case of Aquamid removal. A 33-year-old woman, who had undergone forehead augmentation 12 years previously with an Aquamid injection, visited the department of plastic and reconstructive surgery of our medical center due to a severe forehead contour irregularity. Removal of 20 mL of excess gel was performed by direct incision and squeezing under local anesthesia. Our experience shows that Aquamid removal is possible, but should be performed with appropriate surgical precautions.
Fillers for soft-tissue augmentation have been extensively investigated with respect to safety, efficacy, ease of use, longevity, cost, and the possibility of removal [1]. In particular, gel removal after injection is of concern, and many surgeons are deterred from using them by the possible need for extensive surgery should removal be required. Several studies have demonstrated the safety and efficacy of polyacrylamide hydrogel [2], which has been used for soft-tissue augmentation for more than 25 years with excellent results. In 2001, it was awarded CE certification and has been in clinical use since 2000 under the brand name Aquamid (Contura International, Soeborg, Denmark) [1]. However, Aquamid has been reported to have many short-term complications, such as swelling, hematoma, redness, pain, and itching, as well as several long-term complications such as granuloma formation, contour deformation, depression of the operated area, and migration [3]. To avoid long-term complications such as contour deformity and migration, large volumes (>5 mL per injection) should not be used for facial correction [4], because if migration occurs, removing all the injected material and restoring the normal contour can be difficult [3]. Since the removal of a large volume of Aquamid implanted over a decade previously has not been reported, we describe our experience with such a case herein.
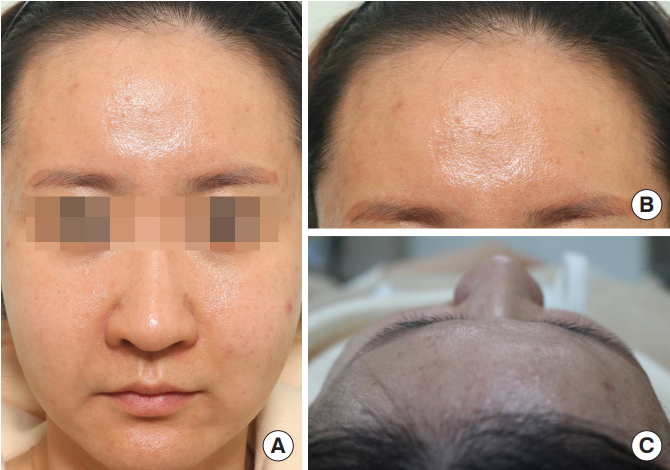
A 33-year-old woman with a forehead contour deformity presented at the Department of Plastic and Reconstructive Surgery of Dongguk University Hospital. She stated that this symptom had appeared 2 years before her visit. She had undergone Aquamid augmentation of the forehead 12 years previously. She vaguely recalled that she had received a sizable amount of filler. However, too much time had passed for her to remember exactly how much filler had been used. Upon presentation, a severe contour irregularity of the forehead was observed, with mild to moderate convexity on the right lateral side and a severe dome-like protruding convexity on the left paramedian side. Both these convexities felt soft and squashy on palpation. The other part of her forehead appeared to be relatively concave (Fig. 1). No tenderness was evident at the convexities.
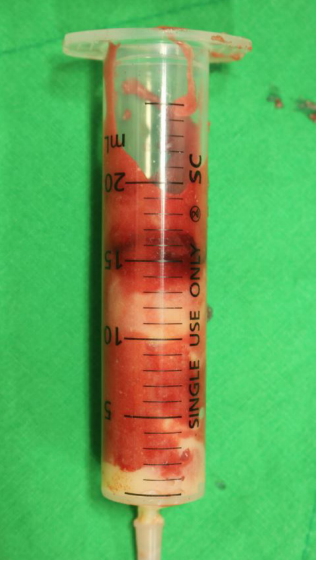
A small amount of filler (0.5 mL) was removed by 18-gauge needle aspiration and a small amount of filler leaked out through the puncture site. Manual squeezing through two 0.5-cm stab incisions under local anesthesia decompressed the forehead convexities (Fig. 2). Finally, 20 mL of Aquamid and blood were removed by squeezing (Fig. 3). The incision sites were repaired with Ethilon 6-0 after thorough saline irrigation. We then administered an oral antibiotic (Meiact, Meiji Seika Pharma, Tokyo, Japan; cefditoren pivoxil; a third-generation cephalosporin) at a dose of 100 mg three times a day to prevent postoperative infection for 5 days.
Sutures were completely removed on postoperative day 5. The patient’s recovery was uneventful and no complications, such as contour deformities or margin irregularities, were evident at 1 month postoperatively (Fig. 4).
Aquamid is used as an injectable soft-tissue filler for facial soft-tissue augmentation and contour enhancement. This filler is a polyacrylamide hydrogel comprised of 2.5% cross-linked polymer and 97.5% sterile water. The gel is colorless and transparent [2] and is usually injected into the nasolabial and glabellar folds, depressed corners of the mouth, and perioral wrinkles. In addition, gel injection is also used for facial contouring, such as for lip augmentation and contouring of the chin, cheeks, nose, and vermilion border [2]. The gel is administered first in the deeper planes of subcutaneous tissues, and then to more superficial layers. Injections directly administered underneath the dermal plane are avoided [2,3].
Aquamid is thought to cause few foreign body reactions because of its physical and chemical properties. The polymer is highly cross-linked and forms strong non-covalent bonds with water, which results in a smooth surface [2]. Nonetheless, Aquamid mediated facial augmentation can have short-term complications of swelling, hematoma, pain, redness, and itching [2], and long-term complications of contour deformation, depression of the operated area, and migration [3]. The causes of polyacrylamide hydrogel migration are various. Gravity can move the gel downward and cause contour deformities, and the fibrous capsules that usually develop around the filler are too flimsy to prevent migration. In addition, facial muscle movements may cause capsule rupture and gel migration [5].
Several cases of injected gel migration have been reported [1,4,5]. As mentioned above, because of its chemical properties, Aquamid is considered to have few side effects, and filler displacement appears to occur roughly 4 years after injection. To the best of our knowledge, this is the first reported case involving the removal of a large volume of gel (20 mL) that had been implanted more than 10 years previously. No consensus has been reached on the treatment of permanent filler–associated complications. Not enough time has passed to assess the long-term efficacy of Aquamid as a clinically safe filling material for soft tissue due to its unstable localization in implant regions and its tendency to move easily in response to gravity and muscle activity [5].
A large number of other synthetic fillers are available in various parts of the world, but it is difficult to comment on the effectiveness and safety of these products because of a lack of published information. Aquamid is a non-FDA approved synthetic filler that has been approved in Europe and in 40 countries [6]. The ideal characteristics of a filler are that it be non-toxic, biocompatible, long-lasting, reversible, autologous, safe, predictable, and not be discernible by touch or by eye, with minimal downtime. Aquamid offers satisfactory permanence, but as with other synthetic fillers, its use has been associated with complications such as granuloma, acute and delayed infections, migration, displacement, and deformities [1-4,6].
In the Korea, the Ministry of Food and Drug Safety provides information on various fillers that have been authorized. This information includes the side effects of synthetic fillers like Aquamid. It is noted that Aquamid is long-lasting but hard to remove, and the Ministry of Food and Drug Safety therefore limits the use of this filler except for improvement of facial wrinkles [7]. It can be helpful for patients to search for information on specific fillers before undergoing procedures.
The development of numerous fillers has provided plastic surgeons with invaluable means of conducting minimally invasive facial rejuvenation. Properly performing procedures and using suitable fillers are the keystones for achieving a natural appearance and avoiding iatrogenic problems. Nevertheless, all fillers have possible side effects, and if a side effect is encountered, a proper work-up and diagnosis followed by appropriate management are essential. In our patient, a large amount of Aquamid was removed with adequate surgical precautions, more than a decade after administration. We have included clinical photographs of the entire procedure. If the filler remains present and contour irregularity persists, an additional imaging work-up for localization followed by surgical removal or needle aspiration can be recommended.
Notes
Ethical approval
The study was performed in accordance with the principles of the Declaration of Helsinki.
Patient consent
The patient provided written informed consent for the publication and the use of her images.
Fig. 1.
(A, B) Forehead contour irregularity in the region where Aquamid had been injected 12 years previously. (C) Mild to moderate convexity was observed on the right lateral side of the forehead and a severe dome-like protruding convexity was observed on the left paramedian side.

REFERENCES
1. Pallua N, Wolter TP. A 5-year assessment of safety and aesthetic results after facial soft-tissue augmentation with polyacrylamide hydrogel (Aquamid): a prospective multicenter study of 251 patients. Plast Reconstr Surg 2010;125:1797-804.


2. von Buelow S, Pallua N. Efficacy and safety of polyacrylamide hydrogel for facial soft-tissue augmentation in a 2-year follow-up: a prospective multicenter study for evaluation of safety and aesthetic results in 101 patients. Plast Reconstr Surg 2006;118(3 Suppl): 85S-91S.


3. Ono S, Ogawa R, Hyakusoku H. Complications after polyacrylamide hydrogel injection for soft-tissue augmentation. Plast Reconstr Surg 2010;126:1349-57.


4. Breiting V, Aasted A, Jorgensen A, et al. A study on patients treated with polyacrylamide hydrogel injection for facial corrections. Aesthetic Plast Surg 2004;28:45-53.


5. Cheng NX, Xu SL, Deng H, et al. Migration of implants: a problem with injectable polyacrylamide gel in aesthetic plastic surgery. Aesthetic Plast Surg 2006;30:215-25.


6. Born TM, Airan LE, Suissa D. Injectables and resurfacing techniques: soft-tissue fillers. In: Rubin JP, Neligan PC, editors. Plastic surgery. 4th ed. Vol. 2. London: Elsevier; 2018. p. 39. -54.
7. Ministry of Food and Drug Safety. A guide for safety use of plastic filler [Internet]. Cheongju: National Institute of Food and Drug Safety Evaluation; c2015.[cited 2019 Apr 22]. Available from: https://www.mfds.go.kr/brd/m_227/view.do?seq=19404.