 |
 |
- Search
| Arch Aesthetic Plast Surg > Volume 30(1); 2024 > Article |
|
Abstract
Background
Local flap techniques are commonly employed for nipple reconstruction. This study was conducted to present the surgical outcomes of composite nipple grafts performed in patients for whom local flap techniques were not suitable.
Methods
This study included 26 patients who underwent composite nipple grafting between February 2014 and March 2021. Data regarding demographics and complications were obtained through retrospective chart review. Complications, including wound dehiscence and total or partial necrosis, were evaluated by the surgeon. The BREAST-Q survey (as described by Spear et al.) was utilized to investigate patient satisfaction with the reconstructed nipple as well as donor-site morbidity.
Results
No instances of wound dehiscence, infection, implant exposure, or total necrosis were noted. However, partial necrosis was observed in five patients (19.2%) and healed well with local wound care. The odds ratio (OR) for partial necrosis was not significantly higher in patients who received radiotherapy than in those who did not (OR=1.06, P=0.97). The color and projection of the reconstructed nipple were generally found to be satisfactory. Regarding donor-site morbidity, sensation and contractility were well preserved.
Conclusions
This study demonstrated that composite nipple grafting is a viable option, as it reduces the size of the relatively large nipple on the normal side while preserving nipple sensation and contractility. This technique can be applied using a thin skin flap that covers the prosthesis following irradiation or a tissue expansion procedure, with minimal risk of compromising the nipple.
The nipple is a landmark that greatly influences the aesthetic appearance of the breasts. While a relatively simple surgical procedure, nipple reconstruction holds substantial psychological and aesthetic importance for patients, as it can impact overall satisfaction, sexuality, and self-confidence. When planning for nipple reconstruction, it is important to attempt to match the position, size, and height of the newly reconstructed nipple (or neo-nipple) with the opposite side. Nipple reconstruction can be performed several months after breast reconstruction surgery, with the timing depending on the type of reconstruction, the surgeon’s discretion, the presence of complications, and the potential need for postmastectomy chemotherapy or radiotherapy [1].
Numerous techniques have been developed for the reconstruction of the nipple-areolar complex. The concept of nipple-areola transplantation and labial graft was introduced by Adams in the 1940s [2]; since then, various grafts have been utilized, including those from toe pulp, auricular cartilage, and the contralateral nipple. These techniques were designed to establish adequate projection through the provision of soft tissue. Since the 1980s, local flaps have been the most frequently described technique for nipple reconstruction. These include the skate flap, star flap, C-V flap, bell flap, and S-flap [3-9].
Although local flap techniques are widely used in nipple reconstruction, they carry certain limitations. Previous research has shown that local flaps can result in a loss of projection ranging from 45% to 70%, a problem that is particularly pronounced in implant-based reconstruction [10,11]. Consequently, many experts recommend creating a nipple up to twice the size of the desired final volume [12]. However, if the projection of the contralateral nipple exceeds 1 cm or its width surpasses 1.5 cm, a local skin flap may be insufficient or overly tight, making it difficult to achieve the proper nipple size. Furthermore, skin flaps that have been irradiated are not suitable for local flaps due to the high risk of complications [13,14].
In such cases, a composite nipple graft may be beneficial. This technique helps achieve consistent nipple size by decreasing the size of the larger nipple on the opposite side, while ensuring a match in nipple color and texture. This method is also associated with reduced donor-site morbidity [15]. The objective of this study was to report the surgical outcomes and donor-site morbidity of composite nipple grafts at a single institution, as measured by the BREAST-Q survey [16].
We retrospectively reviewed the charts of patients who underwent composite nipple grafting between February 2014 and March 2021. This study was approved by the Institutional Review Board of Yonsei University Severance Hospital (IRB No. 4-2023-0540). The decision to proceed with a composite nipple graft was based on several criteria: the presence of an excessively thin skin flap following irradiation or tissue expansion reconstruction, a nipple diameter larger than 1.5 cm and projection greater than 1.0 cm, and the desire to reduce the size of the nipple on the normal side (Table 1). We did not recommend this procedure to unmarried patients or those planning a pregnancy. Data on demographics, treatment, outcomes, and complications were obtained from the chart review. The demographic data included age, comorbidities, type of reconstruction, time between breast reconstruction and nipple reconstruction, follow-up duration, radiotherapy, and chemotherapy. We calculated the time to complete nipple reconstruction from the date of the final breast reconstruction procedure to the date of nipple reconstruction. Complications including nipple congestion, wound dehiscence, and total or partial necrosis were evaluated by the surgeon. Each patient completed the BREAST-Q survey, as described by Spear et al. [16], to assess satisfaction with the reconstructed nipple and perceptions of the contralateral donor nipple. Patients who did not respond to the survey were contacted via telephone.
The surgical design was executed with the patient in an upright position. The position of the recipient site was determined not only by measuring the distance from the nipple on the normal side to the sternal notch and mid-chest line, but also by considering the optimal position of the breast mound in the reconstructed breast. Once the position of the neo-nipple was confirmed, we planned a so-called vertical bipartition to harvest the inferior half-dome. The recipient nipple site was thinly de-epithelialized using a #15 blade. We then undermined the boundary around the de-epithelialized site to facilitate nipple grafting. After confirming the presence of dermal bleeding at the recipient site, we cleansed the blood with normal saline irrigation to ensure successful grafting. A #11 blade was inserted through the half-base of the nipple while the superior half-dome was pulled upward with a traction suture; then, the nipple was divided. The inferior free graft was positioned over the de-epithelialized area and secured with a simple interrupted suture using black silk 5-0. The donor nipple was folded down and closed with a simple running suture using the same material (Fig. 1). A simple dressing was placed by applying antibiotic ointment and laying fluffy gauze around the nipple to prevent compression. Patients were scheduled to visit the outpatient clinic on postoperative days 1, 3, 7, and 10. Approximately 10 days after surgery, all stitches were removed.
This study included a total of 26 patients, with a median age of 49.15 years (range, 40–69 years) (Fig. 2). Of these patients, six (23.1%) underwent autologous breast reconstruction using abdominal tissue, while the remaining 20 (76.9%) underwent implant-based breast reconstruction. All patients treated with implant-based reconstruction also experienced a period of skin expansion using a tissue expander. The mean follow-up duration, calculated from the date of nipple reconstruction to that of the most recent outpatient clinic visit, was 28.12 months (range, 9.04–75.50 months). The average time to nipple reconstruction was 12.73 months (range, 4.20–25.15 months). Nipple-areolar reconstruction was postponed until the completion of chemotherapy and radiation therapy. Patients who underwent radiation therapy were required to wait at least 6 months after the therapy before undergoing nipple reconstruction (Table 2). Eight patients (30.8%) received radiotherapy, and five patients who had previously undergone nipple reconstruction using a C-V flap experienced gradual nipple flattening and subsequently underwent reoperation using a composite nipple graft. None of the patients experienced nipple flattening, wound dehiscence, infection, implant exposure, or total necrosis. Partial necrosis, defined as a graft loss of less than half, was observed in five cases (19.2%), with one of these patients undergoing radiotherapy. All patients recovered well with local wound care.
When comparing patients who received radiotherapy with those who did not, no statistically significant differences were observed in demographic characteristics, including age, comorbidities, type of reconstruction, and type of cancer. Of the eight patients who received radiotherapy, one (12.5%) experienced partial necrosis. In contrast, four (22.2%) of the 18 patients who did not receive radiotherapy developed partial necrosis. The odds ratio (OR) was 1.06, suggesting that the group who received radiotherapy was 1.06 times more likely to experience partial necrosis than the group who did not. However, this difference was not statistically significant (P=0.97).
Of the 26 patients, 19 (73%) completed the provided survey, which covered their satisfaction with the recipient’s nipple as well as donor-site morbidity. When asked about the reconstructed nipple, 47.4%, 73.7%, and 52.7% of patients expressed that they were either somewhat or very satisfied with the natural appearance, color, and projection of the nipple, respectively (Table 3). In relation to the donor nipple, 78.9% of patients reported that the sensation remained unchanged after surgery, and 89.5% confirmed that the donor nipple exhibited normal or nearly normal contraction. All patients characterized the donor nipples as adequate or almost adequate, and the post-surgery impact of the donor nipple on their femininity or sexuality represented either only a slight decrease or no change. Furthermore, 63.2% of patients indicated that they would probably or definitely undergo the procedure again (Table 4).
Ensuring the appropriate size, location, natural appearance, color, texture, and nipple projection is crucial for achieving aesthetic outcomes in nipple reconstruction. Numerous surgical techniques have been developed to date. However, the optimal method may vary, depending on factors such as the diverse histories of different reconstruction types, the thickness of the skin flap, and the use of postoperative adjuvant radiotherapy.
The most common complication in nipple reconstruction using local flap techniques is the gradual loss of projection. Estimates suggest that the loss of nipple projection ranges from 45% to 75% [10]. This progressive loss is primarily caused by the tension exerted by the breast skin, which pulls the neo-nipple back in line with the breast skin [11]. Notably, implant-based reconstruction tends to yield relatively poor results in terms of projection loss. This is because the internal pressure from the implant tends to stretch and flatten the breast surface over time. Furthermore, the capsules surrounding the implant exert contractile forces, leading to a greater loss of projection (47.9% for implant-based reconstruction) compared to autologous breast reconstruction (31.6%) [17]. Many surgeons advise creating a nipple that is 1.5 to 2 times larger than the desired size to account for this anticipated loss of volume [18,19]. Additionally, skin that has been expanded after tissue expansion is relatively thin and lacks the support required for the reconstructed nipple. Therefore, we performed composite nipple grafts on patients exhibiting thin, expanded skin and a large contralateral nipple. We observed no instances of nipple flattening, wound dehiscence, infection, implant exposure, or total nipple necrosis.
The well-documented risk of irradiating skin, particularly thin skin flaps over implants, has led many surgeons to hesitate when considering nipple reconstruction. One study reported a complication rate of 25% for nipple reconstruction using various local flaps after radiotherapy. These complications included total or partial nipple loss, implant exposure, and removal [13]. Another study identified the irradiation field as a significant risk factor, contributing to an increased postoperative complication rate in nipple reconstruction with local flaps [15]. Consequently, we selected patients who had undergone irradiation and who had a large contralateral nipple for composite nipple grafting. In this study, only one patient (12.5%) of the eight who had received radiotherapy experienced partial skin necrosis, which later healed completely. No statistically significant difference was found between the irradiated and non-irradiated patients in terms of partial necrosis (OR=1.06, P=0.97).
The primary innervation of the nipple-areolar complex is provided by the third and fourth anterior cutaneous branches, as well as the fourth lateral cutaneous branch of the intercostal nerves [20,21]. The anterior cutaneous branches of the third and fourth intercostal nerves follow a superficial path within the subcutaneous tissue, culminating in the superomedial nipple. To preserve these branches, we harvested only the lower half of the donor nipple. Consequently, no loss of sensation at the donor site was reported.
Patient satisfaction regarding both the reconstructed and donor nipples was assessed using the BREAST-Q survey described by Spear et al. [16]. Of the patients surveyed, 73.7% and 52.7% expressed being somewhat or very satisfied with the color and projection of the neo-nipple. In comparison, Lee et al. [22] reported satisfaction rates of 59% and 48% for color and projection, respectively. However, satisfaction with the appearance and naturalness of the reconstructed nipple was relatively low, with only 47.4% of patients expressing satisfaction. We hypothesize that these results may be lower than actual satisfaction levels, as patients likely did not fully distinguish between their assessment of the reconstructed nipple and their overall evaluation of the reconstructed breast mound or areola color.
The sensation in the donor nipple was well preserved, as demonstrated by 78.9% of patients who reported no change in sensation following surgery. This finding aligns with the 76% figure described by Lee et al. [22]. Additionally, 73.7% of our patients reported that the donor nipple responded normally to stimulation, a result similar to the 72.4% value reported by Lee et al. [22]. Notably, none of the patients reported a reduction in the projection of the donor nipple. Furthermore, 84.2% of patients reported that the role of the donor nipple in sexual arousal remained the same. These encouraging outcomes regarding the donor nipple following composite nipple grafting may help to alleviate concerns about pain, numbness, or disfigurement among patients who are hesitant to undergo this procedure.
This study did have certain limitations, including a small sample size, the absence of a control group, and reliance on subjective evaluations from patients. However, the findings suggest that composite nipple grafting is a viable approach. This method not only reduces the size of the comparatively large nipple on the normal side, but also preserves nipple sensation and contractility. Furthermore, this technique can be utilized on the thin skin flap that covers the prosthesis following radiation therapy or a tissue expansion procedure, with minimal risk of compromising the nipple.
Notes
Ethical approval
The study was approved by the of Yonsei University Severance Hospital (IRB No. 4-2023-0540) and performed in accordance with the principles of the Declaration of Helsinki.
Patient consent
The patients provided written informed consent for the publication and use of their images.
Fig. 1.
Preoperative and intraoperative photographs of the composite nipple graft procedure. (A) Preoperative photograph of the donor site, showing the frontal view of the incision line. (B) Preoperative photograph of the donor site, depicting the lateral view. (C) Postoperative photograph of the donor site, showing the frontal view after primary closure. (D) Postoperative photograph of the donor site, presenting the inferior oblique view after primary closure. (E) Preoperative photograph of the recipient site, depicting the frontal view after de-epithelialization. (F) Postoperative photograph of the recipient site, showing the frontal view after suturing of the composite nipple. (G) Postoperative photograph of the recipient site, showing the inferior oblique view after primary closure. (H) Postoperative photograph of the bilateral breast, depicting the frontal view.
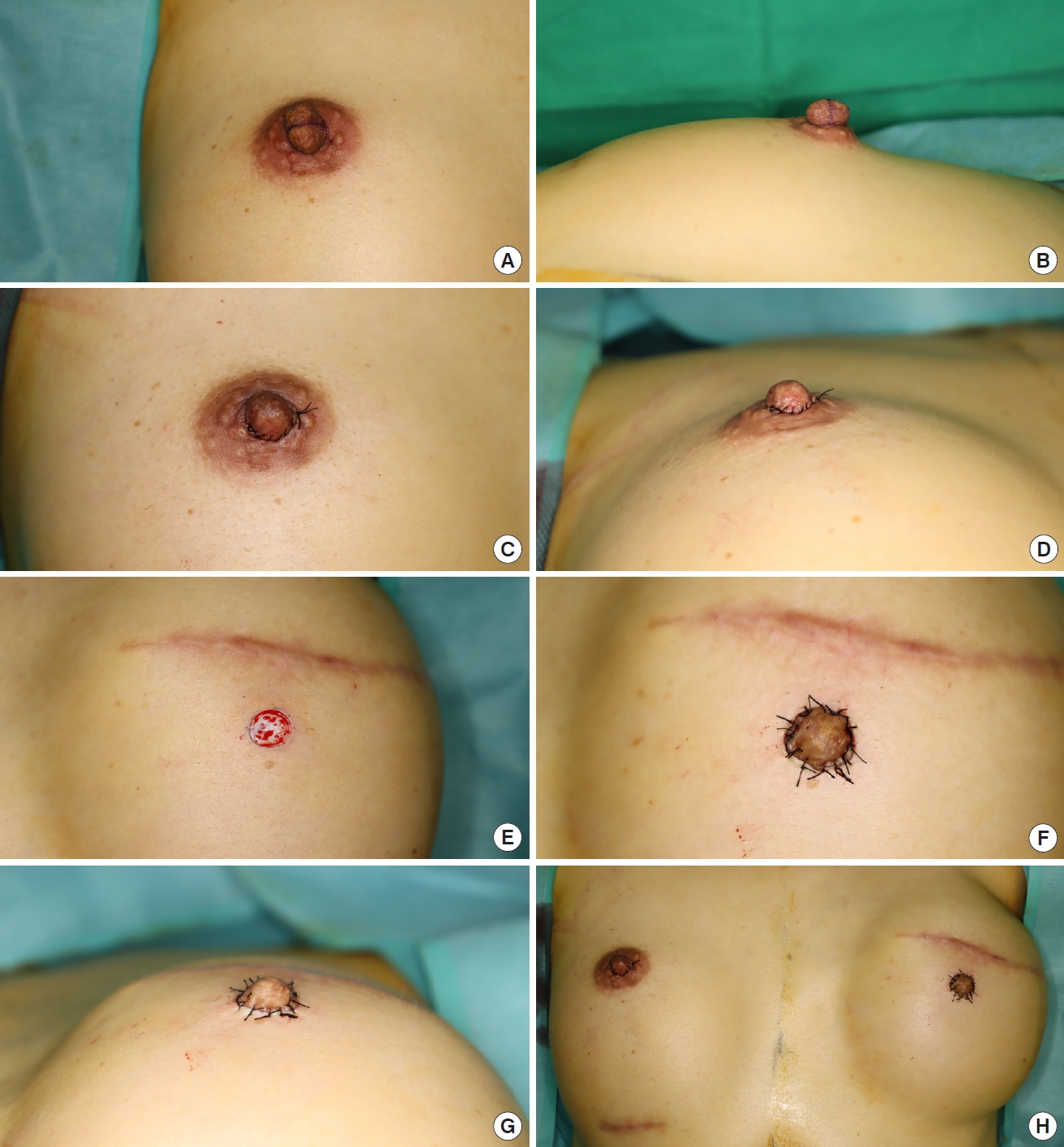
Fig. 2.
A 51-year-old woman underwent a left mastectomy with immediate breast reconstruction using a deep inferior epigastric artery perforator-free flap. Six months later, the patient received a left composite nipple reconstruction. Photographs were captured 1 year after composite nipple reconstruction. (A) Left lateral view. (B) Frontal view. (C) Right lateral view.
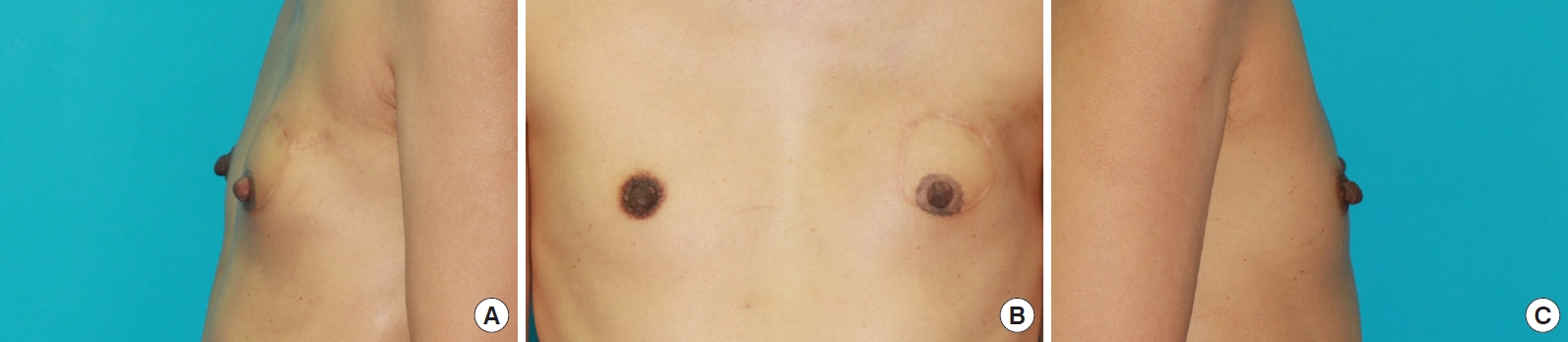
Table 1.
Patient selection for composite nipple graft
Table 2.
Patient demographics
Table 3.
Patient satisfaction with composite nipple graft using the BREAST-Q survey
Table 4.
Donor-site morbidity assessment after surgery using the BREAST-Q survey
REFERENCES
1. Farhadi J, Maksvytyte GK, Schaefer DJ, et al. Reconstruction of the nipple-areola complex: an update. J Plast Reconstr Aesthet Surg 2006;59:40-53.


2. Adams WM. Free transplantation of the nipples and areolae. Surgery 1944;15:186-95.
3. Millard DR Jr. Nipple and areola reconstruction by split-skin graft from the normal side. Plast Reconstr Surg 1972;50:350-3.


4. Brent B, Bostwick J. Nipple-areola reconstruction with auricular tissues. Plast Reconstr Surg 1977;60:353-61.


5. Broadbent TR, Woolf RM, Metz PS. Restoring the mammary areola by a skin graft from the upper inner thigh. Br J Plast Surg 1977;30:220-2.


6. Klatsky SA, Manson PN. Toe pulp free grafts in nipple reconstruction. Plast Reconstr Surg 1981;68:245-8.


7. Hallock GG, Altobelli JA. Cylindrical nipple reconstruction using an H flap. Ann Plast Surg 1993;30:23-6.


8. Little JW 3rd, Munasifi T, McCulloch DT. One-stage reconstruction of a projecting nipple: the quadrapod flap. Plast Reconstr Surg 1983;71:126-33.


10. Di Benedetto G, Sperti V, Pierangeli M, et al. A simple and reliable method of nipple reconstruction using a spiral flap made of residual scar tissue. Plast Reconstr Surg 2004;114:158-61.


11. Krogsgaard SH, Carstensen LF, Thomsen JB, et al. Nipple reconstruction: a novel triple flap design. Plast Reconstr Surg Glob Open 2019;7:e2262.



12. Sodre P, Tobar JS, Vieira R, et al. Nipple areolar reconstruction: techniques to achieve a good result. Mastology 2018;28:119-24.

13. Draper LB, Bui DT, Chiu ES, et al. Nipple-areola reconstruction following chest-wall irradiation for breast cancer: is it safe? Ann Plast Surg 2005;55:12-5.

14. Momeni A, Ghaly M, Gupta D, et al. Nipple reconstruction: risk factors and complications after 189 procedures. Eur J Plast Surg 2013;36:633-8.



15. Amarante JT, Santa-Comba A, Reis J, et al. Halux pulp composite graft in nipple reconstruction. Aesthetic Plast Surg 1994;18:299-300.


16. Spear SL, Schaffner AD, Jespersen MR, et al. Donor-site morbidity and patient satisfaction using a composite nipple graft for unilateral nipple reconstruction in the radiated and nonradiated breast. Plast Reconstr Surg 2011;127:1437-46.


17. Yang CE, Park KH, Lew DH, et al. Dimensional changes in reconstructed nipples: autologous versus prosthetic breast reconstruction. Ann Surg Treat Res 2019;96:8-13.



18. White CP, Gdalevitch P, Strazar R, et al. Surgical tips: areolar tattoo prior to nipple reconstruction. J Plast Reconstr Aesthet Surg 2011;64:1724-6.


19. Banducci DR, Le TK, Hughes KC. Long-term follow-up of a modified Anton-Hartrampf nipple reconstruction. Ann Plast Surg 1999;43:467-70.


20. Schlenz I, Kuzbari R, Gruber H, et al. The sensitivity of the nipple-areola complex: an anatomic study. Plast Reconstr Surg 2000;105:905-9.









